 Take the two structural isomers octane and 2,4-dimethylhexane for example. With few exceptions the more branching that occurs the lower the melting point of the compound. The strength of these forces is determined by the size of the molecule, functional groups present, symmetry of molecular structure and the degree of branching. For molecular substances these forces are namely dipole-dipole bonding, hydrogen bonding, van der Walls dispersion forces. The key factor that influences, viscosity, flash point, boiling and melting points is the type of intermolecular forces acting between molecules and the strength of these forces. 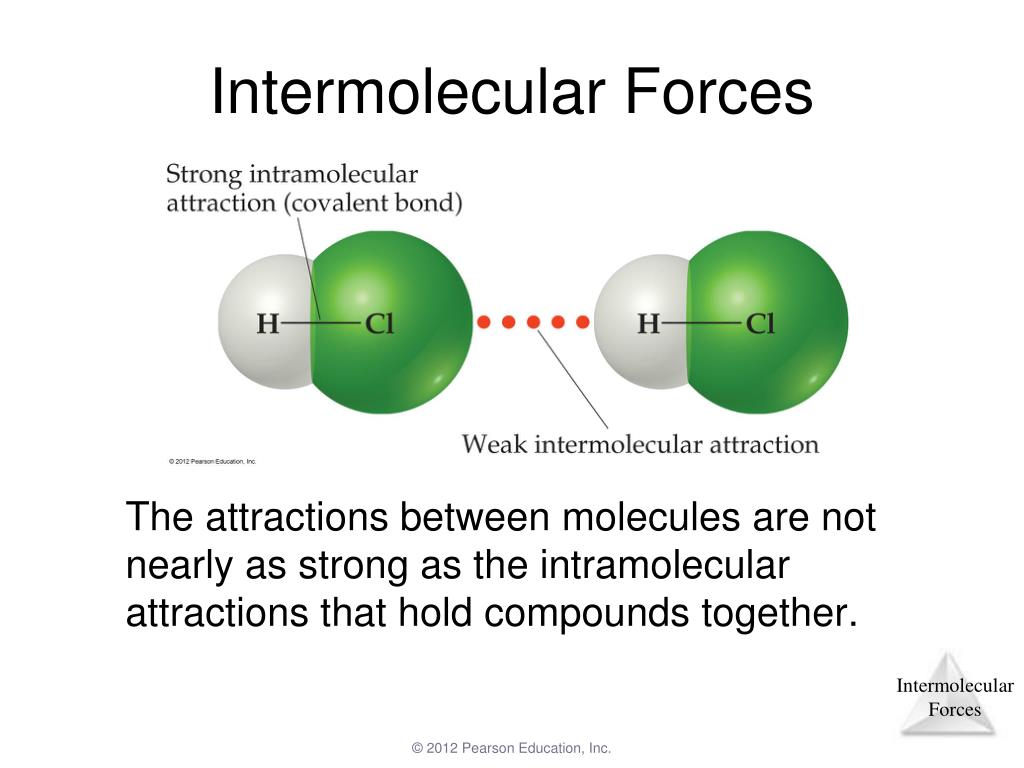
The flash point of a liquid substance is the lowest temperature at which vapours will ignite when an ignition source is applied, such as a flame.īelow is a table of data of some alkanes. 
The greater the resistance, the more viscous a substance is. Viscosity is a measure of how much a liquid resists flowing. When the radii of two atoms differ greatly or are large, their nuclei cannot achieve close proximity when they interact, resulting in a weak interaction.Chemistry -organic molecules-trends in boiling temp, viscosity and flash point Trends-boiling point, viscosity and flash pointīefore we continue let's define two terms, viscosity and flash point. This can account for the relatively low ability of Cl to form hydrogen bonds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
